
ולידציה לנוהל 126 של משרד הבריאות
חברת Smart Logic מומחית בולידציה לנוהל 126 של משרד הבריאות על פי נוהל 126 של משרד הבריאות ישנם תכשירים רבים שעל מנת לשמור על איכותם
סמארט לוג’יק מבצעת תיקי ולידציה למערכות ממוחשבות התואמות את התקנים 21CFRPart11 ו Annex 11 האירופאי.
לייעוץ בנושא ולידציה, התקשרו אלינו 08-910-2070.
בקטגוריה מידע כללי וטכני אודות ולידציה, מאמרים, הדרכות ועוד.

חברת Smart Logic מומחית בולידציה לנוהל 126 של משרד הבריאות על פי נוהל 126 של משרד הבריאות ישנם תכשירים רבים שעל מנת לשמור על איכותם
חברת סמארט לוג’יק מומחית בולידציה לנוהל 126 של משרד הבריאות כמו שראינו במאמר הקודם מהי ולידציה הבנו מה היא ולידציה ומה החשיבות שלה בכל מתקן

Test Incidents- It is important to fully record the details of all new test incidents and maintain an index of these incidents, this article shows you how to analyse, log and classify test Incidents according to GAMP
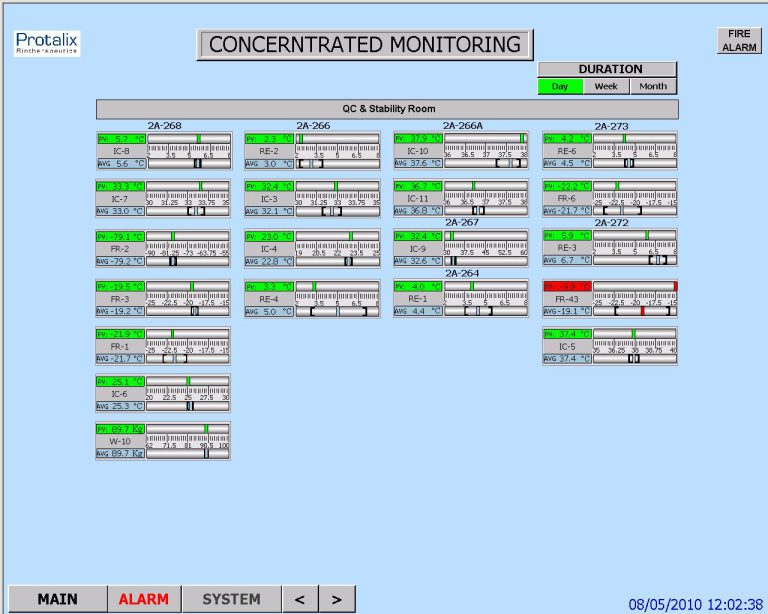
Development life cycle of a system of product containing SW often involves unexpected incidents, predominantly in the testing phase. Whilst defects and failures are unlikely to ever be totally eliminated, occurring incidents should be promptly and correctly dealt with by the test team to minimize the likelihood of passage of avoidable defects intoproduction environment

Schedule of System Operation – SSO Turbine Air Inlet Cooling This article is an elaborated example of a schedule of system operaton – sso we

The third part of our Validation case study includes Documentation for IQ and OQ – to be checked at PDI/FAT, DQ Protocols Including PC/PLC, IQ Protocols Including PC/PLC, OQ Protocols Including PC/PLC, PQ Protocols Including PC/PLC

The second part of our case study deals with the Validation Requirements of Documentation for Initial Tender,Documentation for Design Review,Documentation Prior to FAT or Pre-Delivery Inspection – PDI& Commissioning

This is the first part of Validation Requirements – case study, it covers Document Contents & Responsibility
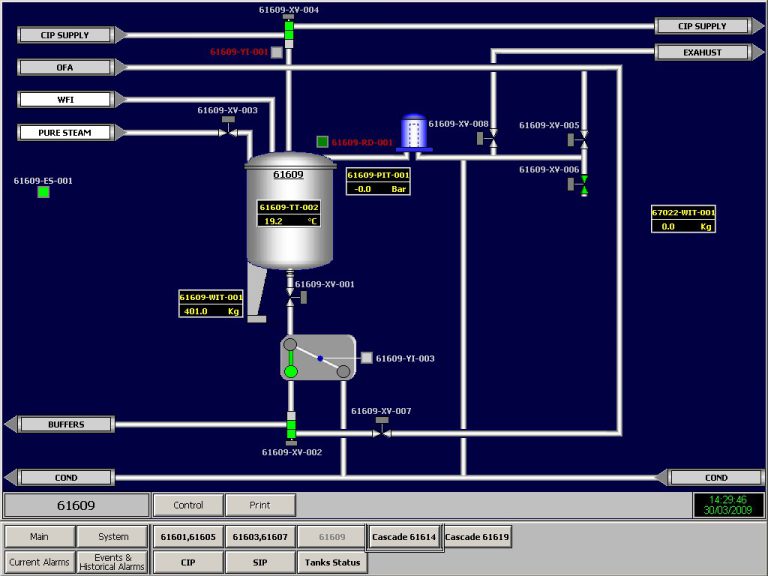
The Function Design Specification (FDS) is part of the validation documentation. In this article I will continue to elaborate the parts of the FSD of system function and the system facilities

ולידציה – Function Design Specification- FDSPreparation Overview

FRS – Regulatory Requirementscover all the FDA specifications regarding the system compliance with the 21 CFR Part 11 definitions, and also with usual validation documentation demandsHMI Requirementsare intended to provide the URS demands from the HMI screens, regarding proper graphic design and functionality for controlling and monitoring the system, as specified in customer’s contract with the supplier

ולידציה -FRS Contents This article was written by Iian Shaya, validation,automation and control expert The FRS presents functional requirements for installing and operating a monitoring