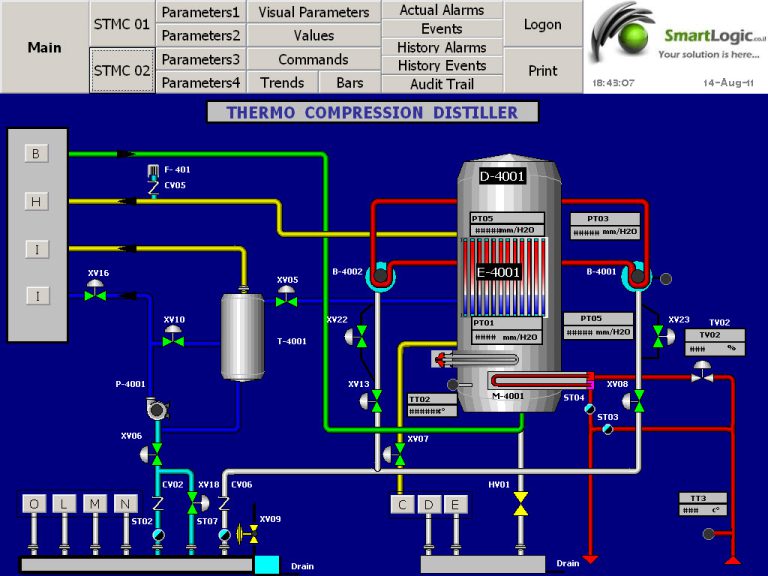
ולידציה – 3 Validation case study – part
The third part of our Validation case study includes Documentation for IQ and OQ – to be checked at PDI/FAT, DQ Protocols Including PC/PLC, IQ Protocols Including PC/PLC, OQ Protocols Including PC/PLC, PQ Protocols Including PC/PLC