ולידציה – URS – Regulatory & HMI Requirements
This article was written by Iian Shaya, validation,automation and control expert
Regulatory Requirements
These requirements cover all the FDA specifications regarding the system compliance with the 21 CFR Part 11 definitions, and also with usual validation documentation demands
Computerized system compliance with 21 CFR Part 11 definitions, such as the system access control by the user's managing personnel, who shall be responsible for the content of the electronic records (ERs) contained in the system
System capability of restricting logical access to the system according to specified authorization levels.
Access to the system allowed only by user ID and a specific password
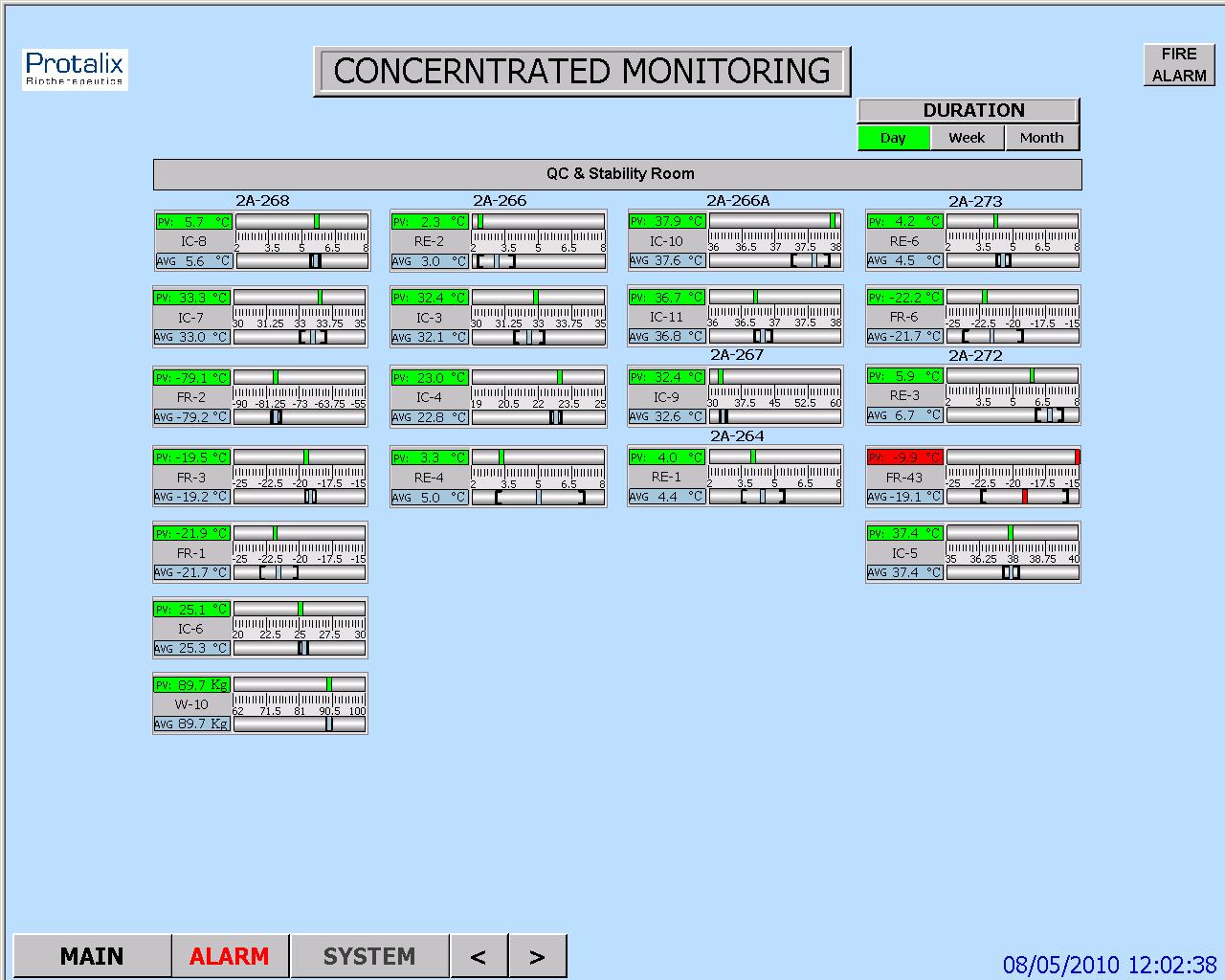
System capability to record the values, alarms, user changes and any other events, and provide readable forms and reports of ER data
Storage of historical events, current alarms and historical alarms data records on the computerized system database
Data display to the user in "view only" mode, so the user cannot alter or delete data/records
Provision of user's capability to backup data daily, weekly and monthly, according to his procedures, to ensure protection of the records and to enable their accurate and ready retrieval throughout the records retention period.
Provision by the supplier of a project plan and quality assurance (QA) processes during development and the testing stages as part of his QA systems
Provision by the supplier of the following documents
Functional Requirements Specification –FRS.
Functional Design Specification – FDS
IO List
Schedule of System Operation – SSO
Installation Qualification (IQ) protocol.
Operation Qualification (OQ) protocol
Performance Qualification (PQ) protoco
HMI Requirements
These requirements cover the provisions demanded from the HMI screens, regarding proper graphic design and functionality for controlling and monitoring the system, as specified in customer's contract with the supplier
:Note
The final contents of the URS and FRS are tailored according to the type and size of the system under validation. Since the URS and FRS regarded herein are generic, they include requirements that may not be necessary in small or simple systems
This article was written by Iian Shaya, validation,automation and control expert